Once again, the periodic table is your BFF. This means we need a conversion factor between grams and moles. How many moles of CH 4 are present in 4.83 g of CH 4?įirst of all, we are given grams of CH 4 and asked to solve for moles of CH 4. Who knew we were drinking so many molecules? Sample ProblemOne of the main components of natural gas is methane (CH 4). Think about that next time you down a glass of water. That means every 1.2 tablespoons of water contains 6.023 x 10¬ 23H 2O molecules. Those 18.02 grams represent one mole of water. Roughly 1.2 tablespoons of water is equal to 18.02 grams. That means 1 mole of water is equal to 18.02 g. Lucky for us (thanks again to the chemistry gnomes), its molar mass is 18.02 g.
We just calculated the molecular mass of water to be 18.02 amu. The molar mass of a compound (in grams) is numerically equal to its molecular mass (in amu). We don't even need a calculator to compute between molecular mass and molar mass…the values are numerically the same. Just as the name suggests the molar mass is the mass (in grams) of one mole of a compound. Molecular mass H 2O = 2(1.008 amu) + 16.00 amu = 18.02 amuįrom the molecular mass we can determine the molar mass of a molecule. We know, we heart the periodic table, too. Where do we find the atomic mass of H and O? Whip out your favorite periodic table. Molecular mass H 2O = 2(atomic mass of H) + atomic mass of O What's the molecular mass of water (H 2O)? To calculate the molecular mass we add together the atomic mass of two hydrogen atoms and one oxygen atom: The molecular mass (sometimes called the molecular weight) is just the sum of the atomic masses in a molecule. We don't want to offend all the atoms out there.) If we know the atomic masses of individual atoms, we can easily calculate the mass of a molecule. Later, after heating, the molecule releases a molecule of carbon dioxide (CO 2).Now that we know all about atomic masses we can move on to bigger and better things…molecules. The organic molecule cyclobutadiene (C 4H 6) combines with propenoic acid (C 3H 4O 2) to form a molecule with the molecular formula of C 7H 10O 2. You have of some unknown transition metal which weighs. Nitrogen and hydrogen: Carbon Dioxide (CO 2) Example Problem Moreover, this means that of NH 3 is equal to of NH 3. For ammonia, we should therefore get a molecular mass of. The last and final step is to add up the two products we got from multiplying. So, now we will multiply these numbers by their corresponding atomic masses. According to the molecular formula (NH 3), there is one nitrogen and three hydrogens present. The second step is to determine how much of each element is present in the compound. For example, if we are trying calculate for ammonia (NH 3), then we need to find the atomic masses for nitrogen and hydrogen. The atomic mass is equal to the atomic number which is listed below the element symbol. The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. Let’s look at some examples of calculating this value for some different molecules.

It should be noted that this number is an average and therefore may vary due to isotopic elements. Molar mass can be calculated by using the periodic table and following three simple steps. The units of molar mass is grams per mole, often abbreviated. That’s a heck of a lot of molecules, which is why the molecular mass of table salt (sodium chloride) is a respectable 58.44 grams per mole, quite a large handful. Both definitions will give you the same result, they are just in different units.Ī “mole” of a substance is defined as atoms or molecules of that substance. Another definition is the sum of the atomic weights of the atoms that make up a molecule. The definition of molar mass is simply the number of grams that one mole of a substance weighs.
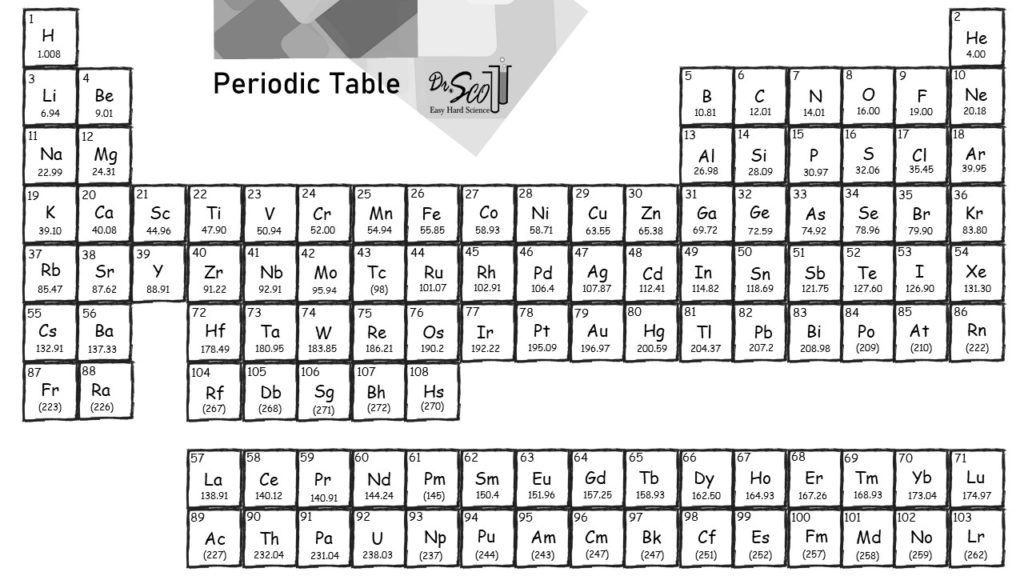
Compounds: Contains atoms of different elements that are combined at a fixed ratio.It is sometimes referred to as molecular weight. Molar mass: The sum of atomic weight in a molecule.Isotope Abundance and Average Atomic Mass.How to Write Electron Shell Configurations.Quantifying Protons, Neutrons, and Electrons.If you enjoy this article, be sure to check out our other tutorials linked below. You will learn how to find and calculate the molar mass for elements and molecules.

In this tutorial, we will explain “what is molar mass”.
